 Even though the weather may have decided that sunshine is to be reserved until an unspecified date later in the year, there are still plenty of other things around and about to make a Kat happy. If these fail, there are still anti-depressants… All of which brings us, in a roundabout way, to the decision of Mr Justice Floyd in Lundbeck v Infosint [2011] EWHC 907 (Pat) handed down yesterday.
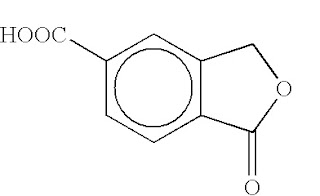
Even though the weather may have decided that sunshine is to be reserved until an unspecified date later in the year, there are still plenty of other things around and about to make a Kat happy. If these fail, there are still anti-depressants… All of which brings us, in a roundabout way, to the decision of Mr Justice Floyd in Lundbeck v Infosint [2011] EWHC 907 (Pat) handed down yesterday.Lundbeck sought the revocation of European Patent (UK) 1 118 614, with priority date of 18 January 2000, relating to a method of making 5-carboxyphthalide (5-cbx) (above), an intermediate compound in the production of the anti-depressant citalopram. The patent had been assigned by Norpharma to Infosint in July 2002. However, owing to a failure to register the transaction at the UKIPO (more on this later) Norpharma remained the registered proprietor until June 2010. For its part, Infosint counterclaimed alleging that Lundbeck was infringing the patent.
Two of the patent’s claims were in issue: claim 1, relating to a process for the preparation of 5-cbx “in an open and however not pressurised reactor”, using “fuming sulfuric acid containing 20-33% by weight of SO3”, amongst other things, and “heating the mixture at 120-145ºC”; and claim 22, of independent validity, relating to “A process for the synthesis of citalopram, in which a process for the synthesis of [5-cbx] according to claim 1 is contained.”
 Various issues of construction were raised, of these the Kat refers to just one in any detail: the claim required the reagents to be “heated at” 120-145ºC. Infosint argued that this range would not be understood by the skilled reader to be exact and would therefore cover temperatures “a few degrees higher than 145ºC.” Lundbeck, for its part, took a narrower view.
Various issues of construction were raised, of these the Kat refers to just one in any detail: the claim required the reagents to be “heated at” 120-145ºC. Infosint argued that this range would not be understood by the skilled reader to be exact and would therefore cover temperatures “a few degrees higher than 145ºC.” Lundbeck, for its part, took a narrower view.When reading this part of the judgment, this Kat was reminded of the discussion in Chef America Inc. v Lamb Weston Inc. 358 F.3d 1371 (Fed. Cir. 2004), in which the court was urged to read a claim requiring the heating of dough to a temperature of between 400ºF and 850ºF, as if it required heating at a temperature in the required range. Despite it being apparent that heating dough to 850ºF would have resulted in “something that … resembles a charcoal briquette” (p.1373), the request was denied on the basis that the court could not redraft the claim even if the only reasonable construction thereof rendered it nonsensical. The Kat notes that it’s often the smallest of words that cause the biggest of problems. However, back to the case in hand:
The Judge explained that he did not think there was:
[43] “…any difficulty about what the numerical upper limit of the claim actually means: it means 145ºC. As it is expressed as a whole number, it probably covers 145.4ºC as well. Apart from that, I cannot see any basis on which it can sensibly be argued that 145ºC means some higher temperature.”There was, however, a subsidiary point namely:
[44] “…whether “heating at” the specified temperature range means that the temperature has to be maintained within the prescribed range for the whole or substantially the whole of the reaction period and if not, for how much of it.”Floyd J concluded that not all of the heating needed to be within the range, noting (at [45]) that he had been persuaded that the alternative conclusion was probably verging on the kind of meticulous verbal analysis against which stern warnings have been made. The skilled person, he explained, would realise that provided he made some 5-cbx within the specified temperature range (ignoring the warming up period) then he would infringe, even if the rest of the 5-cbx was made at a temperature outside the claimed range.
The patent survived an attack on its novelty. The Judge expressed some disquiet (see esp. [89] and [94]) over the TBA decision in T_666/89 UNILEVER/Washing Composition on the validity of so-called “selection” patents, but noted that his conclusions on the disclosure made by the prior art meant that he did not have to express any firm opinion on the correctness or otherwise of the EPO Board’s decision.
The patent was not so fortunate when it came to inventive step. The prior art consisted primarily of two documents: Forney 1 and Forney 2. Forney 1 was a short note appearing in the Journal of Organic Chemistry which disclosed a process of making 5-cbx “cleanly and in excellent yield” by reacting terephthalic acid with formaldehyde in sulfur trioxide media over a range of reaction conditions. Forney 2, which cross referred to Forney 1 on a number of occasions, was a much longer contribution and dealt with the same reaction in more detail.
The questions before the court related essentially to whether the skilled person would find it obvious to adjust the levels of SO3 in the prior art to those claimed in the patent, and whether he would use an open and unpressurised reactor. To cut a long story short, Floyd J considered that both were to be answered in the affirmative.
[109] “Armed with a proper understanding of the papers the skilled person would naturally reach for the sort of oleum [(a mixture of SO3 and sulphuric acid (H2SO4))] he is likely to have at his disposal, which is likely to be in the 20-30% range. Although he might try to obtain a more concentrated oleum, that would not detract from the fact that a 30% oleum was a natural choice.”Furthermore, even though it had been argued that there was no expectation based on the prior art that a process which used 20-33% oleum in an open and unpressurised reactor would achieve results that were as good as those reported in the Forney papers, the Judge nevertheless considered that it would be obvious to adopt these reaction conditions. He explained (at [119] to [120]) that there was “no limitation in the claim about either yield or purity”, and accordingly it was not possible to “approach obviousness on the basis that the claimed process is better, or even as good as Forney’s preferred SO3 process. It is simply another set of process conditions for performing the same reaction.”
The claim was invalid for obviousness.
Turning to claim 22, the use of the claim 1 process in a process of making citalopram, Floyd J explained that a skilled person seeking to make citalopram from 5-cbx under the instructions found in Lundbeck’s own prior art patent application (“513” published May 1998) would need a method for making the 5-cbx. The addressee would be led to perform a search for methods of making 5-cbx and would uncover the Forney work. The obvious consequence of this was that he would use the claimed process to make the intermediate, and the rest of 513 to make citalopram. Claim 22 was therefore obvious in light of 513 and Forney.
Having concluded that the claims were invalid, Floyd J did not strictly need to consider the other arguments, however for the sake of completeness (and in case he was wrong on the obviousness point) he dealt with them anyway. Accordingly, claim 22 was not considered to be insufficient: it was claimed in entirely general terms. As such, it would have been enabled provided it taught one method by which to make citalopram from 5-cbx. There was no suggestion that the skilled person would have encountered any difficulty in doing so.
In respect of Infosint’s claims of infringement, most of the allegedly infringing acts were carried out in relation to processes that fell outside of the claims as construed by the Judge. Most of those that did not were alleged to receive the benefit of the defence under s64 PA 1977 relating to prior use.
The s64 point:
Infosint accepted that Lundbeck had a prior user defence under s64 in relation to citalopram made in accordance with two processes (one carried out in the UK since 1995 and another in Denmark since 1986) that would otherwise have been infringing. This was agreed to extend to importation of citalopram made by those processes. However, Lundbeck also argued that it could claim the defence in relation to the importation of product made from 5-cbx using an amended process (the “two-pot process”) which was designed to avoid the claims of the patent but which, it was acknowledged, ultimately failed to do. Rejecting this argument, Floyd J noted that:
[169] “Mr Trickett was responsible for introducing the two-pot process at LUPUK. He did so under instructions to change the process for patent infringement reasons. He accepted in cross-examination that making the changes was like starting again. The new process (this is what Mr Trickett called it) involved extra equipment, new timings, a pre-heating step and new pipework. Dr Scott described the new process as “operationally different”.
[170] In my judgment Lundbeck are not entitled to a section 64 defence in relation to the importation of citalopram made by two-pot processes. Such processes are not substantially the same as what was done before. To the extent that those processes use the claimed process parameters, they infringe the claims.”Lundbeck’s argument that the importation of escitalopram (one of the enantiomers of citalopram, and the subject of the dispute in this little line of cases) could be saved by the s64 defence was also kicked into the long grass by the Judge:
[172] “…The importation of escitalopram is not substantially the same act as the importation of citalopram…. [A]lthough escitalopram is contained within racemic citalopram, the two materials have different properties, and are different articles of commerce as a result.”The s68 Point
There was also an issue that arose under s68 concerning the registration (or rather the lack thereof) of an assignment of the patent. Readers will be aware that s68 in its original form specified that the consequence of a failure to register the assignment of a patent was that the assignee was to be denied damages or an account of profits for infringements occurring before the transaction was registered. This was amended following the Intellectual Property (Enforcement, etc) Regulations 2006 (SI 2006/1028) removing references to damages and accounts and substituting them with costs and expenses. In this case, the patentee (Norpharma) had assigned their rights in the patent to Infosint in July 2002. The transaction was registered at the EPO the same month. However, despite instructing an agent to attend to the “formalities for recordal” in the UK, the transaction was not recorded on the UK register until June 2010, some 8 years later.
Infosint argued that the registration of the assignment at the EPO should count as a registration for the purposes of s68. Mr Justice Floyd rejected this argument. Given an application to the EPO fragments into a bundle of national rights upon grant, each of which is independently assignable, it would not be reasonable to require the public to consult the EPO’s register “to see whether by any chance an assignment of the patent was registered in the opposition period”. The national registers were clearly where the public’s attention should be directed when seeking the holder of the patent. Furthermore, the fact that the assignee’s agent had failed to register the assignment even though instructed to do so did not mean that the assignee could rely on the safe harbour of s68(b) PA 1977. This provision, the Judge explained, was concerned with whether it was practicable to register the assignment. It clearly was; the agent had simply failed to do so. Accordingly, if there was any infringement, the s68 defence would have succeeded.
Well, says the Kat, having lost its patent, Infosint may need to take some of Lundbeck's medicine...